Antibodies are lifesaving biomolecules – major effectors of vaccines efficacy and powerful therapeutics for cancer treatment. In natural immunity, highly potent antibodies are produced by the descendants of a single antigen-specific B cell after complex processes of cellular and molecular maturation. In the aim of designing new therapies for challenging infections and cancers, our laboratory gathers expertise in cellular immunology, single-cell genomics, and bioinformatics, to decode B cell responses in health and disease.
Overview
Modeling germinal centers through integrative single-cell analyses
High affinity antibody responses to pathogens and vaccines require affinity maturation of antigen-specific B cells in germinal centers (GC). Within the GC microenvironment, GC B cells undergo iterative cycles of proliferation and B cell receptor (BCR) genes somatic hypermutation (SHM) followed by affinity-based selection of high affinity BCR expressing cells. During the transient GC reaction, some GC B cells exit the GC and differentiate into two types of long-lived progeny that are essential for antibody-based immune protection: long-lived antibody-producing plasma cells and memory B cells. One of the aims of our laboratory is to decipher and model the triggers and molecular pathways driving GC B cell differentiation and exit.
Our project relies on integrative single-cell analysis of B cells (parallel analysis of phenotype, BCR mutations, and transcriptome) in mouse and human GC B cells and their progeny, in order to produce high resolution single-cell models of transcriptional changes in GC B cells, and track differentiation and exit of GC B cells to long-lived progeny. In the long run, we aim at integrating a comprehensive map of phenotypic and gene expression changes within a model of GC B cells dynamics and differentiation.
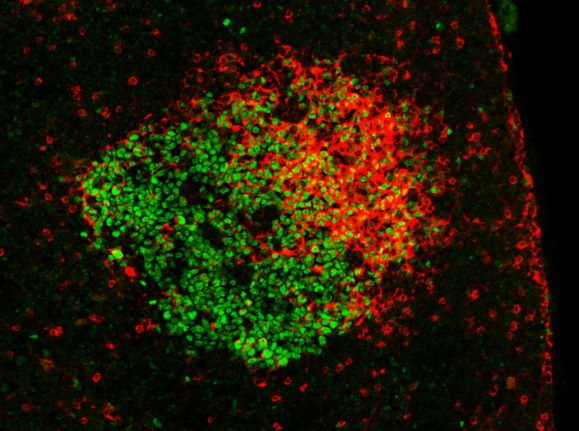
Germinal center in a mouse lymph node after immunization with a model antigen. GC B cells express the BCL6 transcription factor (green). Antigen-specific antibodies (expressed on B cells as BCR or captured on the follicular dendritic cell network) contain a lambda 1 light chain (red).
Copyright Pierre Milpied/ CIML
When B cells go bad: studying cellular heterogeneity in B cell lymphomas
The molecular processes of BCR affinity maturation are vital for host immunity, yet they come with the risk of generating off-target DNA mutations in B cells potentially leading to their cancerous transformation. Indeed, most adult B cell lymphomas originate from GC B cells. Despite considerable progress, B cell lymphomas remain deadly diseases with many patients experiencing relapse and transformation to more aggressive and therapy-refractory tumors. Because bulk analyses are not adapted to unravel the high clonal heterogeneity and clonal dynamics of individual cells composing tumor bulks, we rely on single-cell analysis of human lymphoma samples to study molecular programs in cancerous B cells and other cells of the tumor microenvironment.
Recently, we compared the gene expression profiles of single B cells from follicular lymphoma – a frequent GC B cell derived lymphoma – with those of non-cancerous B cells, and showed that lymphoma B cells diverged consistently from their putative GC B cell-of-origin. In particular, genes which were expressed synchronously in GC B cell states were no longer synchronized in lymphoma B cells. Moreover, we showed that within a single tumor, lymphoma B cells could be engaged in several defined functions or states which suggest that lymphoma B cells have functional plasticity. In collaboration with the Nadel & Roulland team at CIML and the Carnot Institute on Lymphoma (CALYM), our laboratory is further exploring functional cellular heterogeneity in B cell lymphoma using state-of-the-art single-cell genomics and bioinformatics approaches.
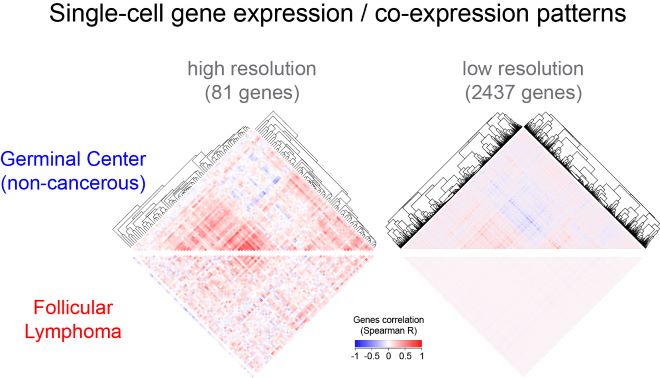
De-synchronization of GC gene expression programs in B cell lymphoma. Clustered gene correlation heatmaps of single-cell gene expression highlight characteristic patterns of genes co-expression in GC B cells (top). Those gene correlation patterns are lost in lymphoma B cells (bottom). The phenomenon which we call “de-synchronization”, can only be observed through single-cell analysis at high (single-cell qRT-PCR) or low resolution (single-cell RNAseq).
Copyright Pierre Milpied / CIML
Decoding human cancer-infiltrating B cell responses
In the fight against cancer, stimulating or re-booting naturally occurring anti-tumor immune responses is an efficient and promising approach. So far, much of the effort to understand and harness anti-tumor immunity has focused on T cell responses, and the potential impact of B cell responses has been neglected. In many solid cancers, the presence of cancer-infiltrating B cells in organized structures resembling GCs at the tumor invasive margin is associated with a good prognosis, suggesting that anti-tumor B cell responses may be orchestrated within the tumor. Yet we currently have little insight into the functional behavior or antigen-specificity of cancer-infiltrating B cells in human tumors.
In our laboratory, we apply integrative single-cell analysis on cells sorted from freshly resected human tumors in order to map the diversity of cancer-infiltrating B cell subsets, define checkpoints of cancer-infiltrating B cell maturation and differentiation in intra-tumor GCs, and define the antigen specificity and potency of antibodies derived from in situ B cell responses. Bridging a major gap in the current understanding of anti-tumor immunity, we hope to identify new ways of stimulating pre-existing anti-tumor neutralizing antibody responses, and provide roadmaps to design effective active or passive anti-tumor immunization therapies.
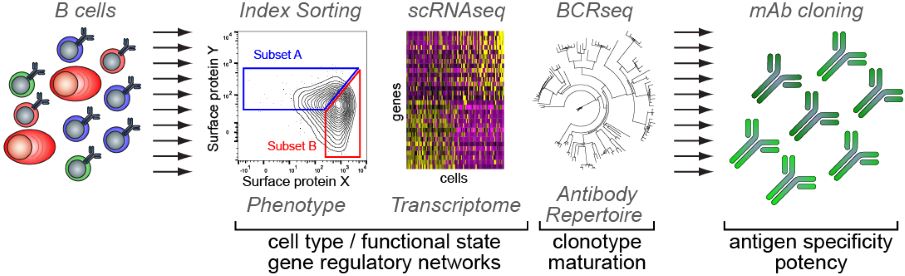
Pipeline for the integrative single-cell analysis of B cells.
Copyright Pierre Milpied / CIML