How do the sentinels of the immune system translate the microbial data they receive into immunological instructions?
In the blood, mucous membranes and lymphoid organs, dendritic cells provide a dual role as sentinels, but also as conductors of the immune orchestra. Hiding in the entryways used by pathogens, dendritic cells locate infectious agents, ingest them and release biochemical signals to alert the first line of defensive cells in the body and draw them to the site of the infection. Once the intruder has been digested, dendritic cells also expose on their surface fragments of the pathogen: the antigens. They then migrate via the lymphatics to secondary lymphoid organs (spleen, lymph nodes, mucosa-associated lymphoid tissue of the digestive tract and lungs) where they present these antigens to T and B lymphocytes. Once "armed" against the pathogen as a consequence of this process, these highly precise defenders in turn migrate to the site of infection to ensure its eradication.
In this sequence of events, the team of Philippe Pierre and Evelina Gatti is particularly interested in the key stage of maturation of dendritic cells. This is the moment when the cells detect the microbes or danger, change their biological functions and begin their migration to secondary lymphoid organs.
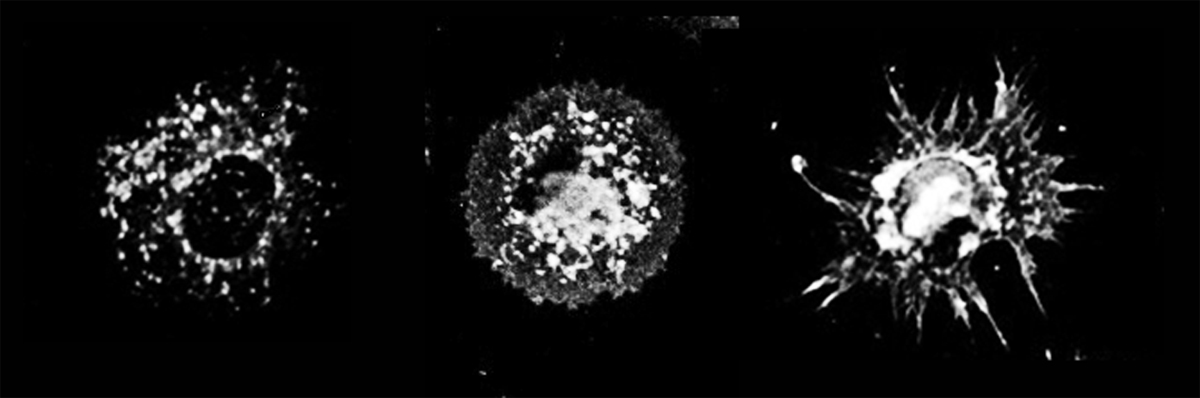 Generation of a dendritic cell. Copyright, Philippe Pierre, CIML
Generation of a dendritic cell. Copyright, Philippe Pierre, CIML
"Dendritic cells work like an interface," recalls Philippe Pierre. "they sense microbial products, classify them according to their type and then convert this information into instructions. They use these instructions not only for themselves - because the encounter with microbial products is not a trivial event and generates natural stress in the cells - but also for immune cells with which they cooperate.
Paradoxically, while we have known for a long time the key role played by these cells in the activation of B and T lymphocytes, and we identified the sensors that allow them to detect danger signals emitted by pathogens, operation of this interface remains partly a mystery. We are trying to solve this puzzle, to understand how the dendritic cells convert "microbial data" into "immunological instructions”.
To deliver these immunological instructions, antigen (in practice a peptide a few amino acids in length) is not presented in isolation to T lymphocytes by dendritic cells but is nestled in a pocket formed by a molecule, known as the Major Histocompatibility Complex (called HLA in humans). MHC proteins determine antigen recognition by lymphocytes and therefore their activation within secondary lymphoid organs. Logically, the team is interested in finding out whether and by what means the dendritic cell redirects the transport of MHC molecules depending on the nature of microbial products that it has detected.
"Under the microscope all dendritic cells change dramatically
in response to microbial products"
"By looking under the microscope at the behavior of MHC molecules in dendritic cells we found that the addition of microbial products induces dramatic changes in all cells in our culture," says Evelina Gatti, co-leader on this team subject. "Initially on the inside of the cells, MHC molecules were suddenly exposed on the outside of the cell. We tried to understand how the dendritic cell organized trafficking of MHC molecules."
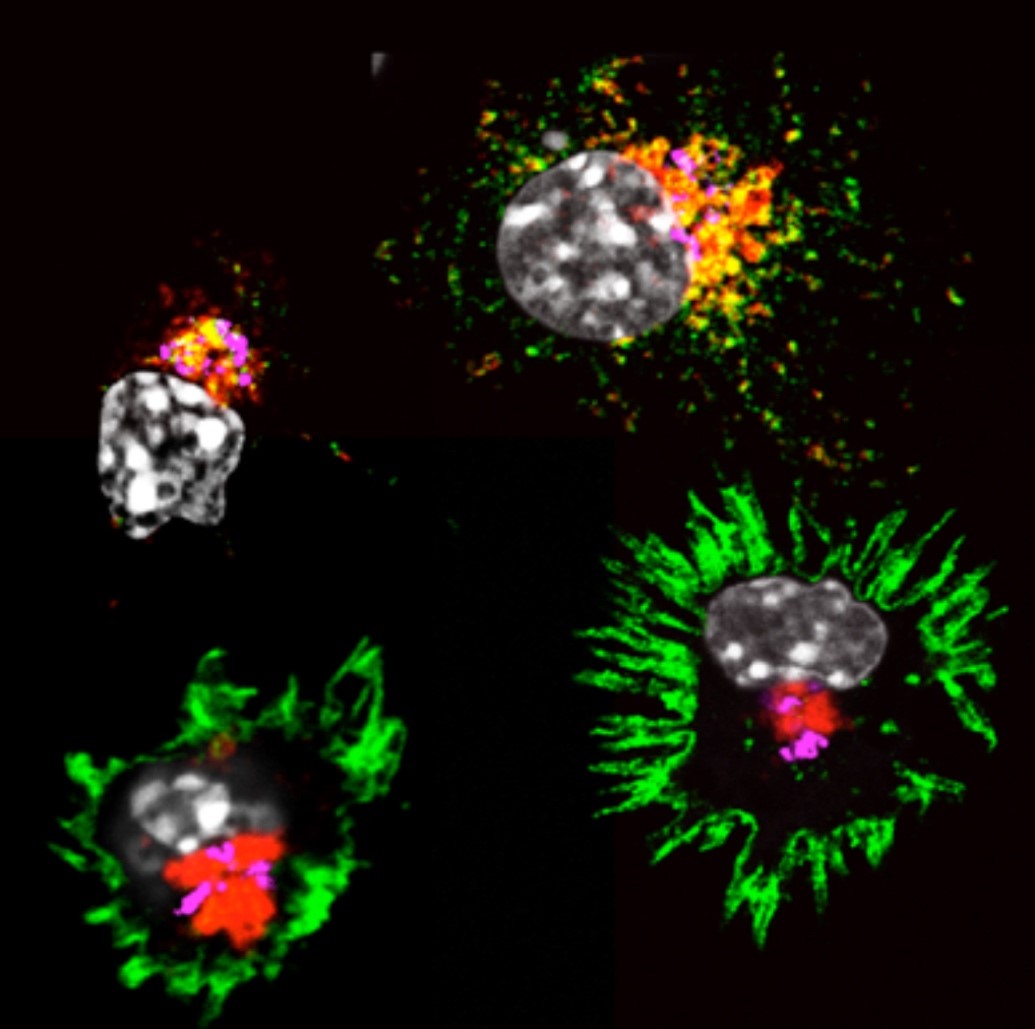
Dendritic cell activation (MHC II molecules in green, Lysosomes in red, nuclei in grey). Picture courtesy of Alexis Combes, CIML
Since these observations, our team has collected an enormous amount of new data on the biochemical pathways involve with the acquisition by dendritic cells of unmatched immunomodulatory functions. While the labeling system of microbial protein was known for many years, the team discovered that enzymes of the ubiquitin ligase family of molecules called MARCH - regulated the routing of MHC molecules during activation of DCs. This allows the molecules to have a "ticket" for restricted access into specialized compartments of the cell. Here the MHC encounter antigens from pathogens or peptides from the self to orient the response of the immune system, either to mount an attack or to signal that they are harmless.
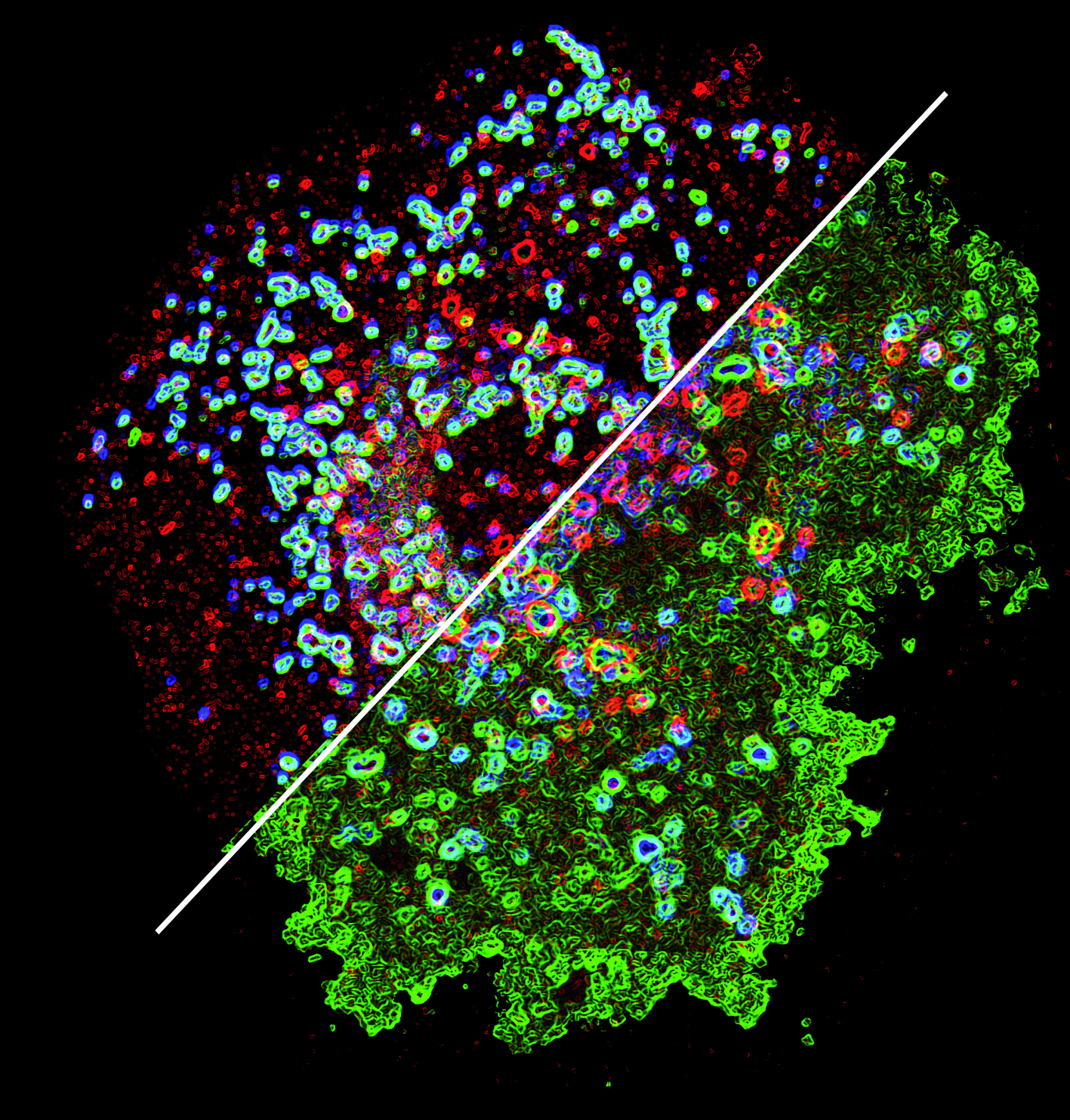
Change in the distribution of MHC class II molecules within a dendritic cell in the presence (top) or absence (bottom) of the ubiquitin ligase MARCH1. Picture courtesy of Aude de Gassart and Evelina Gatti, CIML
Our team was also able to show that brain and DC-associated LAMP associated molecule (BAD-LAMP, C20orf103, LAMP5) is a chaperone for endocytic toll-like receptors (TLR) specifically expressed in the human type-I Interferon producing plasmacytoïd dendritic cells, which are specialized in the detection of nucleic acids potentially of viral or bacterial origin. BAD-LAMP allows the addressing of TLR receptors in intracellular compartments that specifically define the out-put of the signals emitted by these receptors and therefore drives the type of immune response adapted to the detected microbial threat. BAD-LAMP is therefore a key molecule for studying the regulation of human innate immunity in response to nucleic acids and the biology of TLRs that are highly solicited in anti-cancer or anti-viral immunity and often deregulated in a number of autoimmune diseases, like systemic lupus erythematosus (SLE).
At the same time, the team lifted the veil on how protein synthesis, autophagy, and specific molecules that are part of these stress response mechanisms are needed to coordinate the activation of DCs. Several of these molecules (e.g. GADD34) controls the transcription and translational expression of genes encoding key immune mediators or anti-viral effectors like type-I Interferons or TNF.
"In dendritic cell recognition of microbial products, the use of stress pathways and regulation of translation are closely connected. The speed and precision of the response depends on this complex mechanism," point out Philippe Pierre and Evelina Gatti." As a result of these interactions, some molecules are presented on the surface of the cell, some not, others take a different path before being captured by autophagy (a process of self-cannibalism that allows the cell to eliminate excess or damaged proteins)."
The team is now trying to better understand how each of these pathways functions and what are the targets of identified effector molecules. We hope to be able to manipulate these pathways specifically using pharmacology or genetics with the aim of reducing the hyperactivity of the immune system that causes autoimmune diseases or, conversely, to improve the effectiveness of DC stimulation to raise a potent immune response against infectious diseases and importantly cancer.