Our laboratory is studying the physiological mechanisms by which the bone marrow stroma drives lymphoid precursors into functional B lymphocytes, with a special focus on reticular stromal cell populations which control the early steps of B cell differentiation.
The expertise acquired by the analysis of the normal bone narrow microenvironment is applied to pathological situations, as follicular B cell lymphoma. Finally, gene defects responsible for B cell development blockages in primary immune deficiencies, are characterized.
Overview
In the bone marrow (BM), hematopoietic and lymphoid progenitors are present in specialized cellular niches in close contact with mesenchymal stromal cells, indispensable for their differentiation. In this tissue, B cell development is a highly regulated process, which generates pre-BI, pre-BII and immature B cells diversified and tolerant to the self.
Receptors expressed by pre-B cells (the pre-BCR) and B cells (the BCR) act as critical checkpoints, at least by selecting cells carrying functional immunoglobulin (Ig) genes rearranged at the IgH and IgL locus, respectively. Our scientific interests are essentially focused on the physiological mechanisms by which the BM stroma drives lymphoid precursors into functional B cells, and on the defects that affect these processes and lead to pathological situations, aspects largely neglected by the international scientific community, so far.
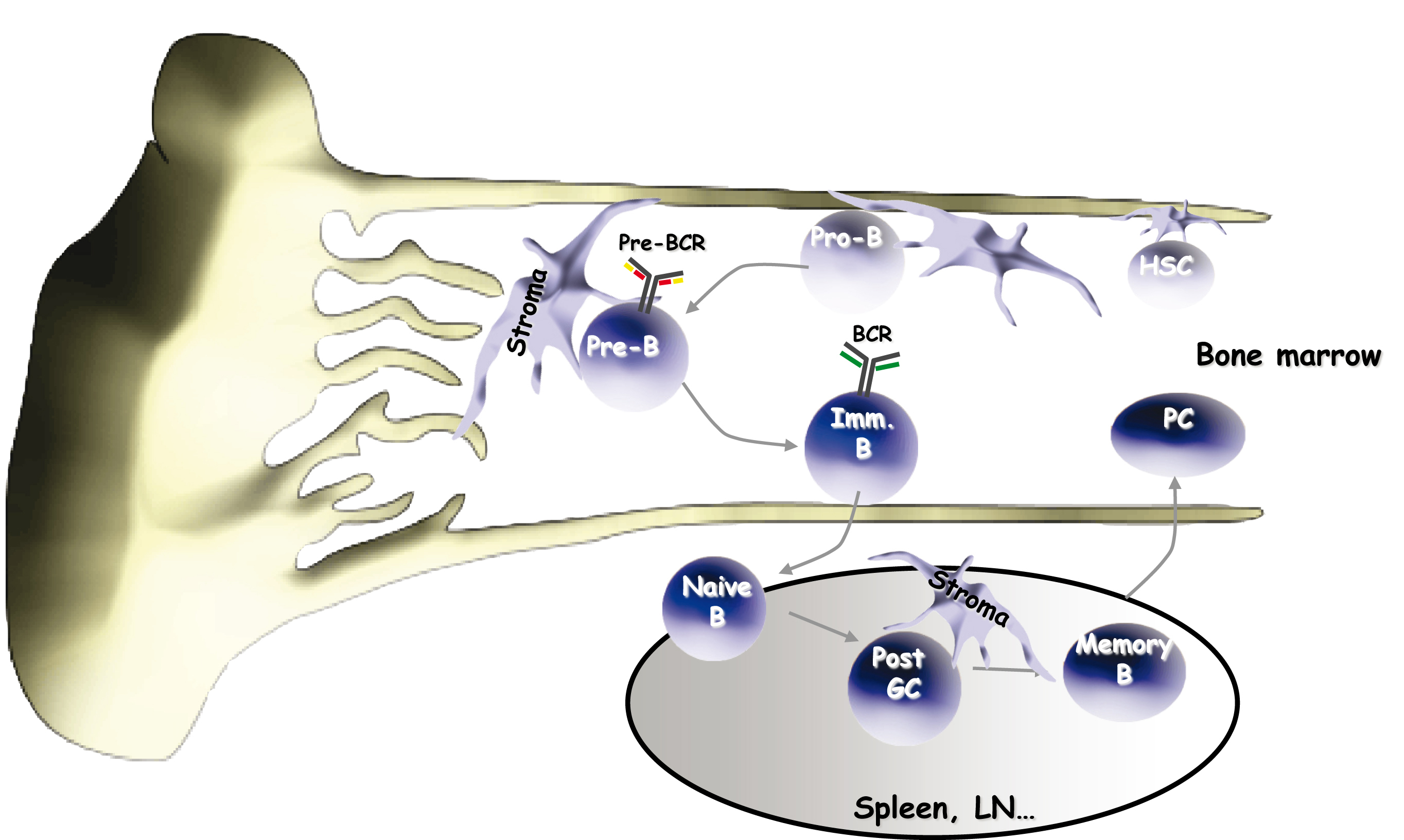
Lympho-stromal interactions during B cell development
Role of the BM stroma in controlling the pre-BCR checkpoint during normal B cell differentiation
Pre-BII cell proliferation and differentiation as well as selection of the Igμ chain repertoire are controlled by the activation of the pre-BCR. This receptor is composed of an Igμ chain, a surrogate light chain (SLC), composed of λ5 and Vpre-B, and the signaling CD79a and CD79b molecules. Our group highly contributed in the understanding of pre-BCR activation, by demonstrating that, in addition to a constitutive signaling, pre-BCR activation was dependent upon a ligand-induced pre-BCR signaling. We identified galectin-1 (GAL1), a member of the S-lectin family, as a pre-BCR ligand, produced by BM stromal cells. We showed that GAL1 creates physical links between pre-B cell integrins and pre-BCRs during formation of pre-BII/stromal cells "synapses".
The structural basis of protein interactions between the pre-BCR and GAL1 are under investigation by Nuclear Magnetic Resonance. Functionally, we demonstrated that crosslinking of pre-BCRs, which leads to their activation, results in efficient proliferation and differentiation of pre-BII cells, in vivo.
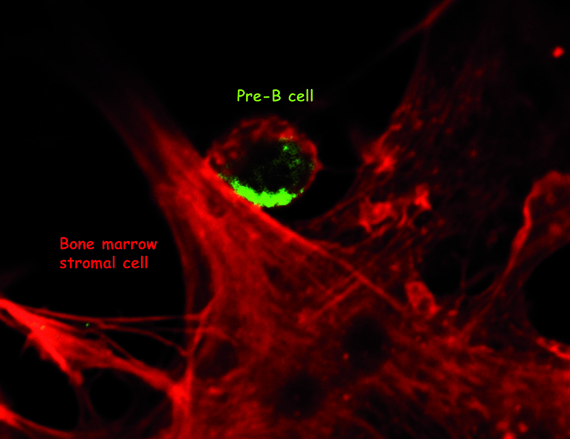
The pre-B cells (in green) are localized in specialized “niches” in the marrow composed of stromal cells (in red). Copyright, Claudine Schiff, CIML
Recent reports indicate that the spatial repartition of stromal cells and hematopoietic progenitors in the BM, follows a strict and coordinated regulation: hematopoietic stem cells (HSC) are in contact with osteoblasts or endothelial cells and move near CXCL12+ reticular cells, whereas early B-lymphocytes migrate from CXCL12+ to IL-7+ cellular niches during differentiation. We recently determined that GAL1 expressing stromal cells are different from IL7+ cells and constitute a specific niche for normal pre-BII cells.
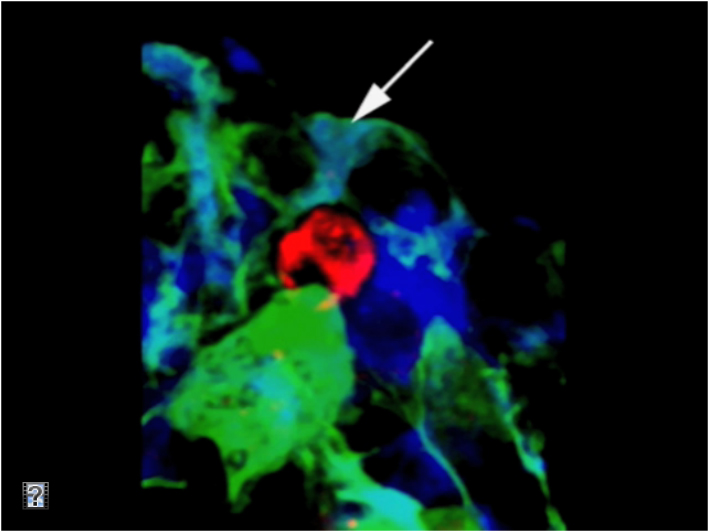
Pre-B cells (in red) contact galectin-1 stromal cells (in green and blue). Copyright, Claudine Schiff, CIML
Finally, identifying the genes responsible for immunodeficiency may help to clarify the regulatory requirements for normal B cell development. In close collaboration with clinicians, we constituted a cohort of rare young patients suffering primary B cell immunodeficiencies, leading to either agammaglobulinemia or hypogammaglobulinemia. We identified two novel gene mutations and are studying their functional effects on B cell differentiation.
Role of the BM stroma in supporting malignant B cell development
In pathology, the BM often provides a preferred microenvironment for tumor cell migration, growth and survival of peripheral B cells. This is the case for follicular B cell lymphoma (FL), which results from the malignant transformation of germinal center (GC) B cells. The knowledge of the cross-talk between FL and follicular dendritic cells in the GC and our competence on the BM microenvironment was the starting point of a collaborative project with B. Nadel, K. Tarte and S. Valitutti aiming at the characterization of BM stromal cells supporting the homing/proliferation of FL and FL precursors. We would like to determine whether FL precursors cells localize in the BM in a pre-existing stromal cell niche or whether they induce their own cellular niche, using mouse and human models.