Projet: HOMEOSTASIS
À propos
Project : HOMEOSTASIS
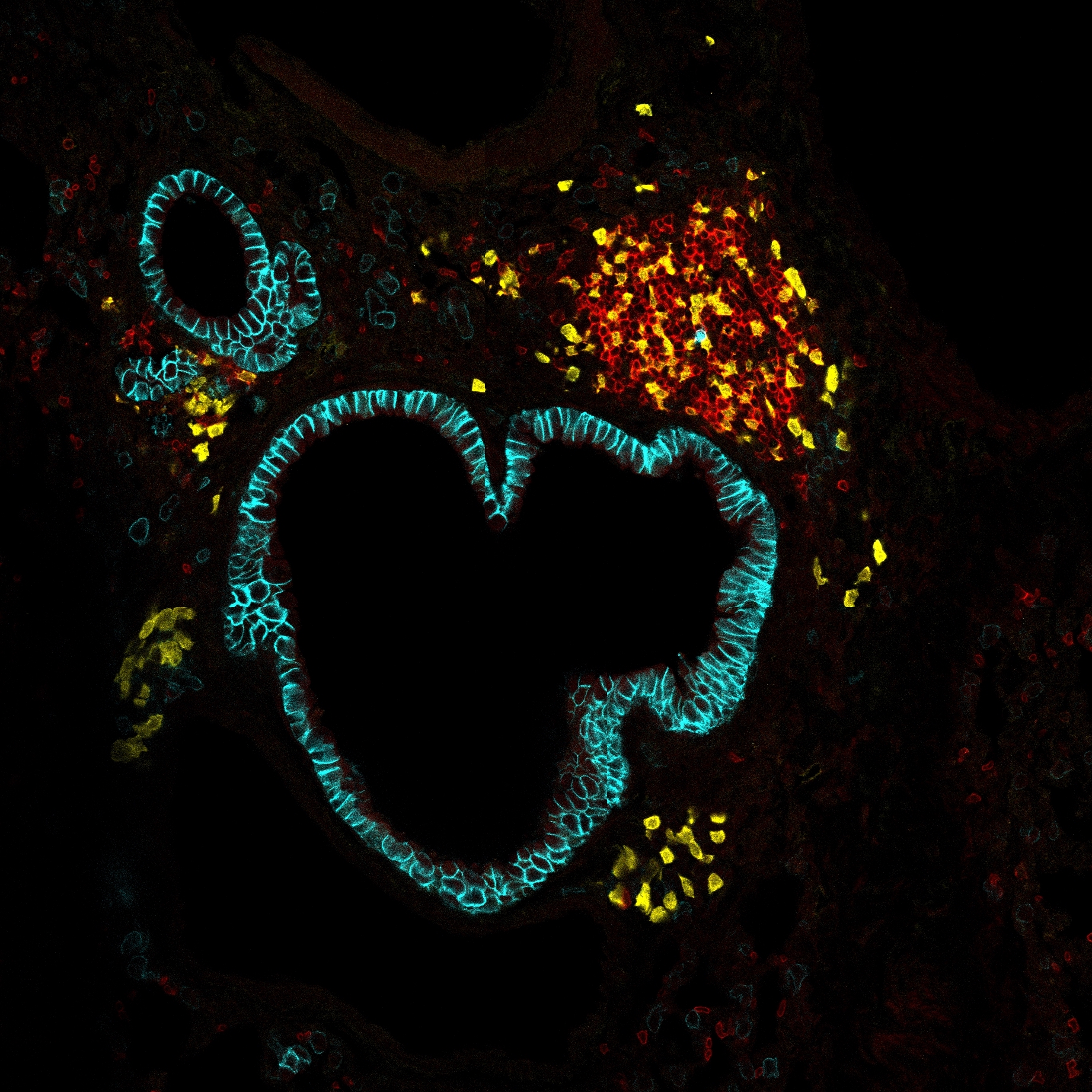
HOMEOSTASIS explores how host–commensal interactions preserve tissue equilibrium. Using a model of long-term, inflammation-free oral colonization by Candida albicans, we investigate the tongue as a key barrier organ. We will (i) characterize myeloid and lymphoid compartments during commensalism, (ii) assess how they support tissue trophic functions, and (iii) define their role in sustaining a stable host–fungus balance. This work will uncover fundamental principles of symbiosis that ensure tissue homeostasis.
.