In the Lego box of the TCR system ... from the list of parts to the assembly instructions!
To fight against infections and tumors, T cells must first be educated. This education takes place in the thymus, a small lymphoid organ located at the base of the neck that grows until puberty and disappears almost completely in adult life. In the thymus the still naive cell learns to differentiate self from nonself and to distinguish a healthy cell from an abnormal cell.
For this purpose, it has on its surface a high precision system of identification: the TCR (T-cell receptor). Using the latter, it recognizes antigen and then integrates and converts the received signal. By a succession of molecular interactions information is transmitted to the nucleus where the cell will make its decision: proliferate, differentiate, migrate or die.
For over 30 years, this extremely sophisticated receptor has been the playground of Marie and Bernard Malissen. After revealing its structure, identifying the molecules that are closely associated with it and having entered into the intimate life of the couple receptor / antigen, the team is now trying to understand the "TCR system" in detail.
Already, by taking apart and putting together the pieces of the puzzle one by one, they discovered a "hinge molecule" whose dysfunction leads to a form of leukemia that has never been described before.
Consisting of two closely associated protein chains, the TCR is the hallmark of T cells. Embedded in its membrane, it recognizes the antigen (in practice a peptide a few amino acids in length) and the self pocket that binds the peptide on the surface of cells specialized in the presentation of the enemy (dendritic cells and macrophages). This molecular complex is a component of a larger protein complex that includes co-receptors and specialized molecules whose function is to deliver signals to the heart of the cell signals triggered by antigen recognition.
When Marie and Bernard Malissen started their career the history of the T cell receptor had just begun. Before becoming world-renowned specialists of the "TCR system" they had to "invent" their object of study!
In the late 70s, scientists were indeed convinced that T cells have a key role in molecular recognition but the nature of this key and the mechanisms leading to cell activation remained to be discovered. Around the world, several research teams then embarked on this adventure. They benefitted from a discovery that proved decisive: the technique of producing monoclonal antibody developed some years earlier by Georges Kohler and Cesar Milstein.
"We broke the system into subsystems and components,
first in cells and then in mice"
"Thanks to the monoclonal antibodies we mapped the identity of the molecules on the surface of T lymphocytes and then tried to block them to measure their impact on cytotoxicity," said Bernard Malissen (some T cells, called helpers boost production of antibodies by B cells while others, known as cytotoxic, destroy the target cell to be eliminated).
To reveal the function of a molecule, the simplest method is to block it with antibodies or even modify or delete it through genetics ... and watch what happens!
"To understand the functioning of many proteins that make up the TCR we acted like the children who receive their first clocks," says Marie Malissen. "First they disassemble them to review all their component parts. Then they try to put them back together. Sometimes they manage to get them working more or less normally, even when parts are missing. At other times they forget a piece and nothing works. Like them we broke the system into subsystems (the transduction module, cell cycle module, the "cytokine factory" module...), first in cultured cells and then in mice using techniques known as site-directed mutagenesis."
With this approach, which prefigured the systems biology currently studied in laboratories around the world, the team reconstructed the cascade of events that allow the T cell to convert the signal coming from the antigenic environment into a multitude of intracellular signals.
This subtle genetic Lego has also recently led to an unexpected discovery. Among the mice developing about 15 different deficiencies in one or the other molecules involved in the signaling cascade, one of them rapidly developed a new form of leukemia!.
"This mouse strain has mutations in a sort of "switch" located just below the TCR. Called LAT (Linker of Activation of T cells), this protein incorporates many signals from the surface and prevents any uncontrolled multiplication of T lymphocytes," says Marie Malissen."Without this brake, T cells proliferate autonomously at breakneck speed (and after them, B cells). Soon, the animals show severe symptoms (fever, cardio-circulatory failure, neurological disorders...) that are very similar to signs found in patients with Chusid syndrome (a disease of unknown etiology also called idiopathic hypereosinophilic syndrome). Thus, the protein LAT is not only a particularly vulnerable "signaling node" but it's also obviously a key factor for the functional balance and the final differentiation of T lymphocytes."
"The relative flexibility of TCR recognition site allows our immune system to catch small viral outbreaks. That's why we avoid most infections."
Alongside this building game, the team has been working on another challenge: determining the three dimensional structure of complex of the T cell receptor, peptide antigen, and a molecule of the self (proteins called the Major Histocompatibility Complex, or MHC, which condition recognition of antigen by T lymphocytes).
"This result, the fruit of ten years working with the team of Dominique Housset at the CEA in Grenoble, was initially a real aesthetic pleasure. We were among the first to observe "in reality" how the T cell receptor recognizes the peptide antigen trapped in the MHC molecule," enthuses Bernard Malissen.
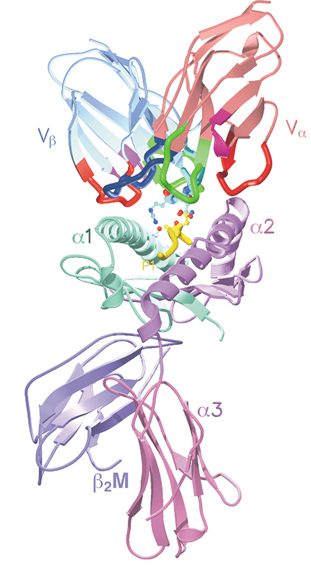
The molecules of the Major Histocompatibility Complex (figured at the bottom of the figure) are present on the surface of dendritic cells. They sample the protein fragments (shown in yellow) contained within these cells. If they are of foreign origin (viruses, bacteria ...), the T-cell receptor (located in the top of the figure) is able to read this information and inform the T cell. Copyright M and B Malissen, CIML.
And then, surprise, whereas the antigen recognition by the TCR is highly selective, its recognition site undergoes structural rearrangements!
"But fortunately these are conformational changes!" exclaims Bernard Malissen. "The recognition site is not totally degenerate, of course, rather it adapts. As an expert in evolution would say, there is evolvability. Ergo: If one partner mutates the other will adjust to this change. The immune system must constantly respond to the variability of the virus. If the relationship were to be unique, a subtle change in the viral surface would be enough to fool the TCR, which in turn would result in the collapse of the entire system. In fact, the relative "flexibility" of the TCR recognition site can often permit our immune system to catch up with small mutations in viruses. That's why we remain free from most infections."
After strolling through structural biology, the team is again examining the lymphoproliferative disease that it has highlighted. The goal? Identify molecules that work with the LAT protein. "In humans, some forms of leukemia may be due not only to defects in this protein brake, but also to failures of one or other molecules that work in concert with it,""says Marie Malissen.
In an attempt to identify the "satellite" of LAT, the team has initiated a reverse genetics approach by chemical mutagenesis in mice. Unlike the classical approach to inactivate a gene previously identified to characterize its function, they pose the question "What are the genes whose mutation affects the function of LAT?" without any a priori hypotheses on the nature of the products of these genes.
Still, 30 years after the beginnings of the exploration of the TCR, this universe comprising a multitude of signals and subtle enzymatic balances, opens rather an infinity of combinations. Beyond this new project, how do Marie and Bernard Malissen intend to deal with this level of complexity?
"Biology is a powerful tool when it comes to splitting a system into its components. That's what allowed us to reconstruct the different pathways of the TCR system. In less than a second, the T cell decodes the packet of information brought by the dendritic cell. Briefly, the TCR starts the process by binding to the couple peptide / MHC and costimulatory molecules and co-inhibitors (CD28, CTLA4...), becomes aware of the context (has the dendritic cell been in contact with bacteria? a virus? tumor?...). How does the T cell integrate the information from these different routes in such a short time? It is still a mystery! Our hypothesis is that there are surely coincidence detectors within every cell of the body. These are capable of determining whether two pathways are activated simultaneously. Still, the integrative dimension is not easily accessible using currently available techniques," concluded Bernard Malissen.
In an attempt to advance on this issue and strengthen the immuno-pathological aspect of their approach, Bernard Malissen will open soon in Marseille the "Centre pour Immunophénomiques" (CIPHE). This new type of institute will first develop, on an accelerated basis, very large-scale mouse models able to mimic, as closely as possible, the real complexity of our immune system.